 |
 |
- Search
| J Neurointensive Care > Volume 6(1); 2023 > Article |
|
Abstract
Background
SARS-CoV-2 induced respiratory illness is increasingly being recognized to be associated with neurological manifestations including an increase in the incidence of strokes, particularly those induced by large vessel occlusion (LVO). Given this, the aim of present study was to determine the influence of SARS-CoV-2 i.e. Coronavirus disease-19 (COVID-19) on mortality, neurological outcomes, and treatment response in patients with stroke due to large vessel occlusion induced by COVID-19.
Methods
A search of randomized controlled trials (RCTs), prospective and retrospective cohort studies was conducted through PUBMED, SCOPUS, MEDLINE, EMBASE, the Central Cochrane Registry of Controlled Trials, and CINAHL databases. The statistical analysis was performed using the relative risk with the Mantel-Haenszel methodology for dichotomous variables with a fixed-effects model. The Newcastle-Ottawa scale (NOS) was used to assess the quality of the publications and ROBINS-I tool was used to evaluate the risk of bias across the studies.
Results
Six retrospective observational cohort and case-control studies involving 1000 patients with LVO were included. The group of COVID 19 patients with LVO had a greater risk of mortality(OR= 7.09, [95% CI: 4.6-10.91], I2= 0%, p ≤0.00001), fewer rates of treatment success(OR 0.15 [95% CI 0.08-0.29], I2 = 49%, p ≤0.00001), and lower favorable outcomes (OR 0.39 [95% CI 0.16-0.96], I2 = 63%, p = 0.04) than COVID 19 negative patients with LVO.
Coronavirus disease-19 (COVID-19) had a global impact due to severe pulmonary involvement. Although infections and fatalities connected with this virus have reduced due to widespread vaccination, however there have been over 518 million confirmed cases and over 6 million deaths worldwide as of May 18, 20221). Even though COVID-19 has been mainly associated with respiratory sysmptoms, many neurological manifestations have been reported2,3); moreover, these manifestations have been linked to greater in-hospital mortality4). One of the main manifestations of this neurological presentation is acute stroke. The study by Misra et al. 5), which included 350 studies, providing data from 145,721 patients with COVID-19, found that one in 50 patients experienced a stroke. In particular, the increase in cases of large vessel occlusion (LVO) in patients with COVID-196) is noteworthy due to the increased risk of mortality and worst sixth-month good outcome (modified Rankin Scale score ≤2) that they present7).We conducted a systematic review and meta-analysis to assess the influence of COVID 19 on clinical outcomes and treatment success in patients with stroke due to occlusion of large vessels.
A detailed search of randomized controlled trials (RCTs), prospective and retrospective cohort studies was conducted through PUBMED, SCOPUS, MEDLINE, EMBASE, the Central Cochrane Registry of Controlled Trials, and CINAHL until April 2022. The search strategy included subject headings (MeSH) and text words connected with Boolean terms, resulting in the following: (“Stroke” [Mesh term] OR “Ischemic stroke[Mesh term]) AND (“Large vessel occlusion” OR “large vessel stroke” OR “intracranial large vessel occlusion” [Mesh term]) AND (“COVID-19” OR “SARS CoV 2 Infection” OR “Coronavirus disease” [Mesh term]) AND (“RTC” OR “randomized clinical trial” OR “observational studies)
The included studies were evaluated based on the following inclusion criteria: (1) RCTs, (2) quasi-RCTs (3) Prospective and retrospective observational studies comparing COVID 19 positive vs. COVID 19 negative patients (control group) with large vessel occlusion stroke.
The quality of the included studies was evaluated using the Newcastle – Ottawa Quality Assessment Scale8). The criteria for determining high, moderate, and low methodological quality were as follows:eight or higher, six to seven, and five or less, respectively. To assess the risk of bias, ROBINS-I was used9).
Individually and separately, the following data were extracted: mortality, functional independence (modified Rankin scale 0 to 2, or Glasgow Prognostic Scale with a score of 4 or greater), treatment success (assessed by the rate of recanalization of occluded vessels), contact was made with the authors for missing data. The doubts were dispelled by consultation and consensus. The statistical analysis was performed using the relative risk with the Mantel-Haenszel methodology for dichotomous variables with a fixed-effects analysis model calculated using the Review Manager 5.3 software. Heterogeneity was determined by calculating Chi-square (I2), with a high level of heterogeneity among the included studies exceeding 65%.
Following a comprehensive search for the information, 115 citations were found; after removing duplicates, screening titles, and abstracts, ten articles were chosen for full-text examination; finally, seven were retained. fourty studies were excluded during the screening phase as they were preclinical studies and case reports, and four were excluded during the full-text examination since two did not evaluate the control group10,11), one was a case report12), and one made another comparison13) (Tables 1 and 2; Fig. 1).
All studies were categorized based on their risk of bias: three had a low risk14-16), and three had a moderate risk17-19) using the ROBINS-I tool. Two of the included studies17,19) exhibited a severe risk of missing data since they did not report the type of intervention for stroke revascularization. Simultaneously, three had a moderate risk in the cofounding domain16-18) and interventions classification14,17,19) (Fig. 2). Regarding the quality assessment, two studies were considered high quality15,19), three were of moderate quality14,16,18), and one was of low quality17). The latter was due to its non-representative sample size of 36 patients, 13 for the COVID group, and lack of a defined intervention protocol. In contrast, missing data could have influenced the outcomes of the studies conducted by Altschul et al., Kihira et al., and Perry et al., since the procedure of thrombolysis or endovascular thrombectomy might have altered the prognosis due to treatment-associated complications that were not recorded (Table 2).
Assessment of publication bias using the Funnel plot revealed asymmetry, consistent with publication bias; however, it was created using less than 10 studies, making the statistical analysis unreliable (Fig. 3).
There was a total of six studies included (Table 3). One thousand participants with LVO stroke were eligible for the meta-analysis; of these, 292 had a positive PCR test for SARS-CoV-2, and 708 had a negative test. For each study, the total patient population, and its division into COVID-19 negative or COVID-19 positive patients with LVO were reported as the type of treatment received. Each study documented mortality and favorable outcomes, while the incidence of complete recanalization was reported in three of these studies.
Mortality was reported in all included studies; 96 (32.87%) of 291 COVID-19 patientswith LVO died, compared to 40 (5.68%) of 706 patients in the COVID-19 negative LVO control group. The pooled estimate revealed a statistically significant difference (p ≤ 0.00001), demonstrating that the presence of LVO associated with COVID-19 increases 7.09 times the mortality compared to the COVID-19 negative LVO control group (OR= 7.09, [95% CI: 4.6-10.91], I2= 0%, Fig. 2).
In the group of COVID-19 patients with LVO, 39 (13.35%) of 292 patients presented a favorable outcome, compared to 380 (53.82%) of 806 patients in the COVID-19 negative LVO control group; thisindicates that the favorable neurological outcome was lower in COVID-19 patients with LVO than the control group (OR 0.15 [95% CI 0.08-0.29] p ≤ 0.00001) with acceptable homogeneity (Chi2 = 9.84, I2 = 49%, Figs. 3-6).
The success of stroke treatment was reported by 4 studies and was measured by the rate of full recanalization following revascularization therapy. 129 (47.75%) of 269 COVID-19 patients with LVO exhibited treatment success, compared to 269 (54.89%) of 490 patients in the COVID-19 negative LVO control group; this indicates that treatment was less effective in COVID-19 patients with LVO than in the control group (OR 0.39 [95% CI 0.16-0.96] p = 0.04) with high heterogeneity (Chi2 = 9.61, I2 = 63%, Fig. 4).
In this systematic review and meta-analysis, we aimed to assess the influence of COVID 19 on clinical outcomes and treatment success in patients with stroke due to occlusion of large vessels. The specific mechanism by which the virus causes strokes is not well known; however, it is thought that there are four processes responsible for its appearance; these include neuroinvasion, endotheliitis, ACE2 suppression, and hypercoagulable state. To begin, the SARS-CoV-2 has been identified in the brain tissue from autopsies of COVID 19 patients20); Song et al. 21). investigated the potential of SARS-CoV-2 to infect the brain using three different techniques; first, it was discovered that infected and adjacent neurons exhibited clear signs of infection and metabolic changes using brain organoids. Second, SARS-CoV-2 neuroinvasion was shown in vivo using mice overexpressing human Angiotensin-converting enzyme 2 (ACE2). Lastly, SARS-CoV-2 was discovered in cortical neurons during autopsies of COVID-19-related deaths. Although the exact mechanism by which neuroinvasion occurs is unclear, two mechanisms have been proposed. The first is through the neuronal pathways, where transneuronal transport of SARS-CoV-2 occurs via peripheral nerve endings22), particularly in the olfactory mucosa23,24). The second mechanism is the hematogenous route; as endothelial cells exhibit SARS-CoV-2 receptors, infection of mucosal linings may provide entry to the lymphatic system and circulation8,25); this route allows the virus to travel to several tissues, including the brain. Regarding endotheliitis, the virus has been found in brain endothelial capillaries of COVID 19 patients during the autopsy; furthermore, Stancu et al. 26). reported the case of an 81-year-old patient with endotheliopathy suggestive of endotheliitis with several strokes; all of this illustrates the virus's inflammatory effect on brain vessels, which can lead to endothelial dysfunction and strokes. This dysfunction is worsened by ACE2 deprivation caused by the virus, which, in conjunction with the hypercoagulable condition, contributes to the stroke's pathophysiology.
Through this study, it was demonstrated that COVID 19 patients with LVO had a greater mortality risk than the control group (p ≤ 0.00001), exhibiting statistical significance. Studies have shown that patients with COVID-19 and stroke had a higher frequency of large vessel occlusion and higher in-hospital mortality rate than stroke patients without COVID-1927).Subsequently, the results showed that favorable outcomes were lower in the COVID 19 patients with LVO than in the control group. This result is supported by Fabregas et al. study28 in which LVO and COVID-19 had a lower likelihood of achieving a favorable functional outcome than those who did not have COVID-19; however, this difference was not statistically significant (p = 0.079).
Regarding treatment success, it was shown that it was less effective in COVID-19 patients with LVO than in the control group 19(OR 0.39 [95% CI 0.16-0.96]; p = 0.04). However, even though sufficient recanalization indicates treatment success, this might result in poor results.In a study conducted by Escalard et al., ten COVID 19 patients with LVO were included, five of these patients got intravenous alteplase, and they all had thrombectomy; although nine of the ten patients had an effective recanalization, none exhibited early neurological improvement, four had early cerebral reocclusion, and six patients died in the hospital29).
Finally, concerning the limitations, we observed that there was a significant disparity in the number of patients in the control group and the COVID 19 patients with LVO, with the latter group having at least twice as many patients. In addition, as previously indicated, some studies did not report the treatment performed, which can cause alterations in the reported results.
In conclusion, this systematic review and meta-analysis demonstrated that patients with LVO had a greater mortality risk, fewer rates of treatment success, and lower favorable outcomes than patients with COVID 19 negative patients with LVO. The findings of this study will serve as a foundation for further research on the subject.
Acknowledgements
We declare that we have no conflicts of interest, and we did not receive financial assistance.
Fig. 1.
Process of study selection. Flow chart of our search strategy and inclusion and exclusion criteria.
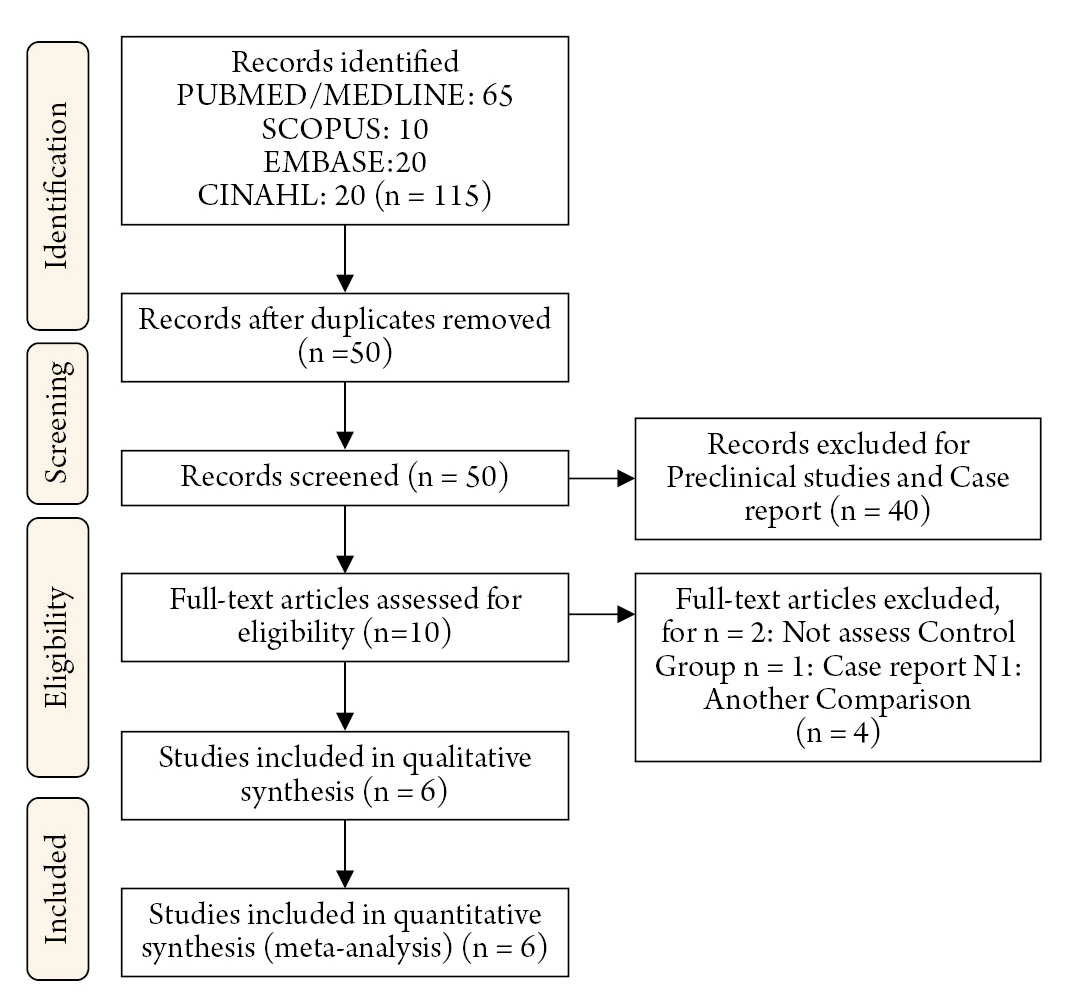
Fig. 2.
Risk of bias (A) risk of bias graph: review authors' judgments about each risk of bias item presented as percentages across all included studies. (B) risk of bias summary: review authors' judgments about each risk of bias item for each included study.
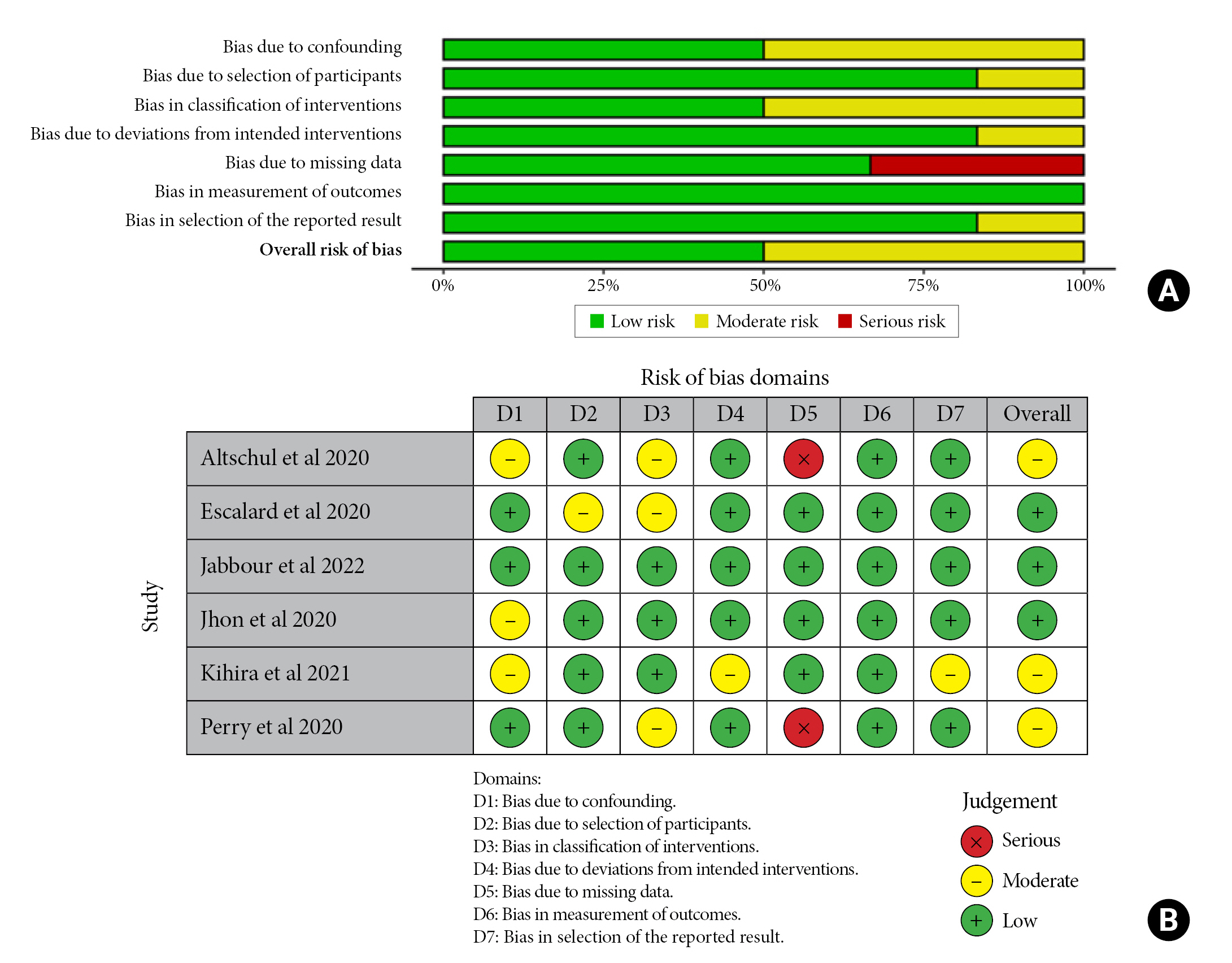
Fig. 3.
Funnel plots. (A) Comparison: 1 Large Occlusion Vessel COVID 19 Positive Vs COVID 19 Negative, outcome: 1.1 Mortality. (B) Comparison: 1 Large Occlusion Vessel COVID 19 Positive Vs COVID 19 Negative, outcome: 1.2 Favorable Outcome. (C) Comparison: 1 Large Occlusion Vessel COVID 19 Positive Vs COVID 19 Negative, outcome: 1.3 Complete recanalization of occluded vessels.

Fig. 4.
Forest plot of comparison: 1 Large Occlusion Vessel COVID 19 Positive Vs COVID 19 Negative, outcome: 1.1 Mortality.
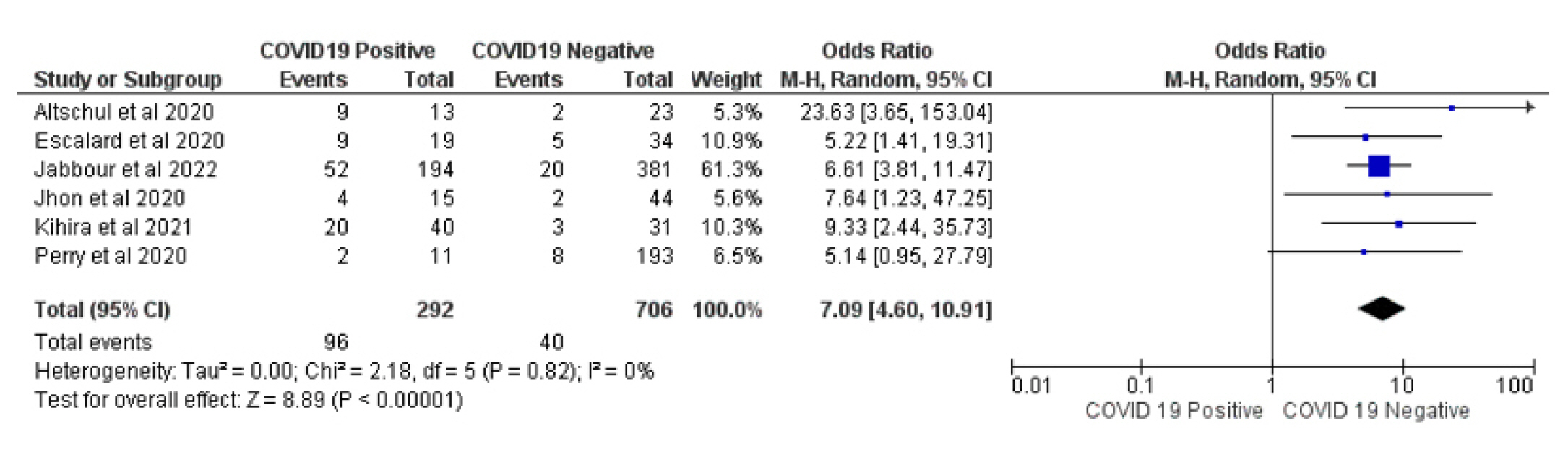
Fig. 5.
Forest plot of comparison: 1 Large Occlusion Vessel COVID 19 Positive Vs COVID 19 Negative, outcome: 1.2 Favorable Outcome.
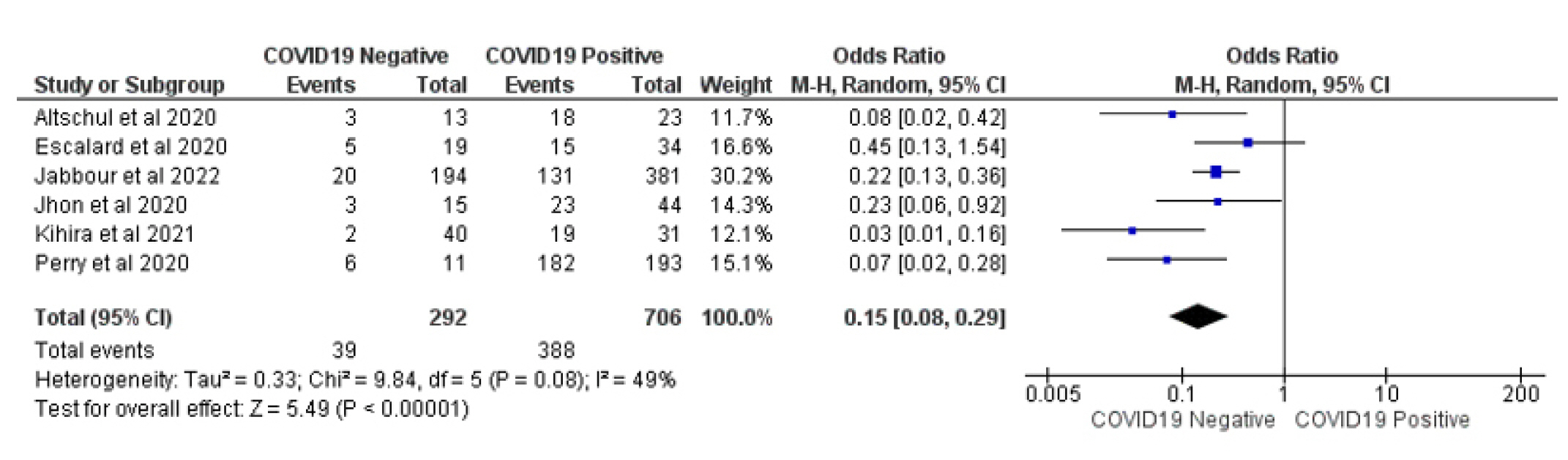
Fig. 6.
Forest plot of comparison: 1 Large Occlusion Vessel COVID 19 Positive Vs COVID 19 Negative, outcome: 1.3 Complete recanalization of occluded vessels.

Table 1.
Excluded studies
| Study | Reason for exclusion |
|---|---|
| Robles et al 202112) | Is a Case Report |
| Majidi et al 202010) | This study only describes an epidemiologic and demographic characteristic of a population without a control group |
| Khandelwal et al 202111) | This study only describes an epidemiologic and demographic characteristic of a population without a control group |
| Cagnazzo et al 202113) | Comparison between Thrombolysis and Mechanical Thrombectomy with an endovascular approach in COVID 19. This study excluded stroke with negative for COVID 19 test. |
Table 2.
Assessment of quality for included studies with New-Casttle Ottawa Scale
| Study | Representativeness of Sample | Size Sample | Source of information | Demonstration that outcome was not present at study start | Confusion variable control | Assessment of outcome | Enough follow-up period | Newcastle Ottawa Scale Score | Quality of study |
|---|---|---|---|---|---|---|---|---|---|
| Altschul et al 202017) | ★ | ★ | ★ | ★ | 4/9 | Low | |||
| Escalard et al 202014) | ★ | ★ | ★ | ★ | ★ | ★ | 6/9 | Moderate | |
| Jabbour et al 202215) | ★★ | ★★ | ★ | ★ | ★ | ★ | ★ | 9/9 | High |
| John et al 202016) | ★ | ★ | ★ | ★ | ★ | ★ | 6/9 | Moderate | |
| Kihira et al 202118) | ★ | ★ | ★ | ★ | ★ | ★ | 6/9 | Moderate | |
| Perry et al 202019) | ★★ | ★★ | ★ | ★ | ★ | ★ | 8/9 | High |
Table 3.
Characteristics of included studies
| Study | Type | Year | Country | N ¶ | Type of treatment (N) | Outcome a ssessed | Length Follow up |
|---|---|---|---|---|---|---|---|
| Altschul et al. 17) | Retrospective Observational Cohort Study | 2020 | USA | Total population | Thrombolysis | - Mortality | 6 Months |
| 36 | 8* | - Favorable outcome (mRS) | |||||
| COVID19 Positive | Thrombectomy | ||||||
| 13 | 12* | ||||||
| COVID19 Negative | |||||||
| 23 | |||||||
| Escalard et al. 14) | Retrospective Observational Cohort Study | 2019 to 2020 | France | Total population | Thrombolysis | - Mortality | 1 year |
| 55 | 25 | - Favorable outcome (mRS) | |||||
| COVID19 Positive | Thrombectomy | - Complete recanalization rate | |||||
| 19 | 30 | ||||||
| COVID19 Negative | |||||||
| 34 | |||||||
| Jabbour et al. 15) | Retrospective Observational Cohort Study | 2020 to 2021 | USA | Total population | Thrombolysis | - Mortality | 6 Months |
| 575 | 267 | - Favorable outcome (mRS) | |||||
| COVID19 Positive | Thrombectomy | - Complete recanalization rate | |||||
| 194 | 308 | ||||||
| COVID19 Negative | |||||||
| 381 | |||||||
| John et al. 16) | Retrospective Observational Cohort Study | 2020 | United Arab Emirates | Total population | Thrombolysis | - Mortality | 6 Months |
| 59 | 34 | - Favorable outcome (mRS) | |||||
| COVID19 Positive | Thrombectomy | - Complete recanalization rate | |||||
| 15 | 25 | ||||||
| COVID19 Negative | |||||||
| 44 | |||||||
| Kihira et al. 18) | Retrospective case-control study | 2020 | USA | Total population | Not reported | - Mortality | 3 Months |
| 71 | - Favorable outcome (mRS) | ||||||
| COVID19 Positive | - Complete recanalization rate | ||||||
| 40 | |||||||
| COVID19 Negative | |||||||
| 31 | |||||||
| Perry et al. 19) | Retrospective case-control study | 2020 | United Kingdom | Total population | Not reported | - Mortality | 1 year |
| 204 | - Favorable outcome (mRS) | ||||||
| COVID19 Positive | |||||||
| 11 | |||||||
| COVID19 Negative | |||||||
| 193 |
REFERENCES
1. Weekly epidemiological update on COVID-19 - 18 May 2022. Accessed May 18, 2022, 2022. https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---18-may-2022.
2. Fraiman P, Godeiro Junior C, Moro E, Cavallieri F, Zedde M. COVID-19 and Cerebrovascular Diseases: A Systematic Review and Perspectives for Stroke Management. Systematic Review. Frontiers in Neurology 2020;11.
3. Tsivgoulis G, Palaiodimou L, Zand R, Lioutas VA, Krogias C, Katsanos AH, et al. COVID-19 and cerebrovascular diseases: a comprehensive overview. Ther Adv Neurol Disord 2020;13:1756286420978004.




4. Chou SH, Beghi E, Helbok R, Moro E, Sampson J, Altamirano V, et al. GCS-NeuroCOVID Consortium and ENERGY Consortium. Global Incidence of Neurological Manifestations Among Patients Hospitalized With COVID-19—A Report for the GCS-NeuroCOVID Consortium and the ENERGY Consortium. JAMA Network Open 2021;4:e2112131–e2112131.


5. Misra S, Kolappa K, Prasad M, Radhakrishnan D, Thakur KT, Solomon T, et al. Frequency of Neurologic Manifestations in COVID-19: A Systematic Review and Meta-analysis. Neurology 2017;97:e2269–e2281.



6. Alhashim A, Alqarni M, Alabdali M, Alshurem M, Albakr A, Hadhiah K. Large vessel occlusion causing cerebral ischemic stroke in previously healthy middle-aged recently recovered from severe COVID-19 infection. Int Med Case Rep J 2021;14:577–582.




7. Smith WS, Lev MH, English JD, Camargo EC, Chou M, Johnston SC. Significance of large vessel intracranial occlusion causing acute ischemic stroke and TIA. Stroke 2009;40:3834–3840.



8. Wells GA, Wells G, Shea B, et al. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. 2014.
9. Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. Bmj 2016;355:i4919.



10. Majidi S, Fifi JT, Ladner TR, Lara-Reyna J, Yaeger KA, Yim B, et al. Emergent Large Vessel Occlusion Stroke During New York City's COVID-19 Outbreak: Clinical Characteristics and Paraclinical Findings. Stroke 2020;51:2656–2663.



11. Khandelwal P, Al-Mufti F, Tiwari A, Singla A, Dmytriw AA, Piano M, et al. Incidence, Characteristics and Outcomes of Large Vessel Stroke in COVID-19 Cohort: An International Multicenter Study. Neurosurgery 2021;89:E35–E41.




12. Robles LA. Bilateral Large Vessel Occlusion Causing Massive Ischemic Stroke in a COVID-19 Patient. J Stroke Cerebrovasc Dis 2021;30:105609.



13. Cagnazzo F, Piotin M, Escalard S, Maier B, Ribo M, Requena M, et al. European Multicenter Study of ET-COVID-19. Stroke 2021;52:31–39.

14. Escalard S, Chalumeau V, Escalard C, Redjem H, Delvoye F, Hébert S, et al. Early brain imaging shows increased severity of acute ischemic strokes with large vessel occlusion in COVID-19 patients. Stroke 2020;51:3366–3370.



15. Jabbour P, Dmytriw AA, Sweid A, Piotin M, Bekelis K, Sourour N, et al. Characteristics of a COVID-19 cohort with large vessel occlusion: a multicenter international study. Neurosurgery 2022;90:725–733.

16. John S, Kesav P, Mifsud VA, Piechowski-Jozwiak B, Dibu J, Bayrlee A, et al. Characteristics of large-vessel occlusion associated with COVID-19 and Ischemic Stroke. AJNR Am J Neuroradiol 2020;41:2263–2268.



17. Altschul DJ, Esenwa C, Haranhalli N, Unda SR, de La Garza Ramos R, Dardick J, et al. Predictors of mortality for patients with COVID-19 and large vessel occlusion. Interv Neuroradiol 2020;26:623–628.




18. Kihira S, Schefflein J, Mahmoudi K, Rigney B, N Delman B, Mocco J, et al. Association of Coronavirus Disease (COVID-19) With large vessel occlusion strokes: a case-control study. AJR Am J Roentgenol 2021;216:150–156.


19. Perry RJ, Smith CJ, Roffe C, Simister R, Narayanamoorthi S, Marigold R, et al. Characteristics and outcomes of COVID-19 associated stroke: a UK multicentre case-control study. J Neurol Neurosurg Psychiatry 2021;92:242–248.


20. Gu J, Gong E, Zhang B, Zheng J, Gao Z, Zhong Y, et al. Multiple organ infection and the pathogenesis of SARS. J Exp Med 2005;202:415–24.




21. Song E, Zhang C, Israelow B, Lu-Culligan A, Prado AV, Skriabine S, et al. Neuroinvasion of SARS-CoV-2 in human and mouse brain. J Exp Med 2021;218:e20202135.



22. Lima M, Siokas V, Aloizou AM, Liampas I, Mentis AA, Tsouris Z, et al. Unraveling the possible routes of SARS-COV-2 invasion into the central nervous system. Curr Treat Options Neurol 2020;22:37.




23. Meinhardt J, Radke J, Dittmayer C, Franz J, Thomas C, Mothes R, et al. Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nature Neuroscience 2021;24:168–175.



24. De Melo GD, Lazarini F, Levallois S, Hautefort C Michel V, Larrous F, et al. COVID-19-associated olfactory dysfunction reveals SARS-CoV-2 neuroinvasion and persistence in the olfactory system. bioRxiv 2020, https://doi.org/10.1101/2020.11.18.388819.
25. Bostancıklıoğlu M. SARS-CoV2 entry and spread in the lymphatic drainage system of the brain. Brain Behav Immun 2020;87:122–123.



26. Stancu P, Uginet M, Assal F, Allali G, Lovblad KO. COVID-19 associated stroke and cerebral endotheliitis. J Neuroradiol 2021;48:291–292.



27. Nannoni S, de Groot R, Bell S, Markus HS. Stroke in COVID-19: a systematic review and meta-analysis. International Journal of Stroke 2021;16:137–149.




- TOOLS






